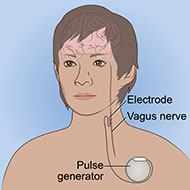

Vagus nerve stimulation (VNS) is a surgical procedure that involves a device implanted under the skin. The device sends electrical pulses through the left vagus nerve that runs from the brainstem through the neck and down the side of the chest and abdomen. The nerve carries messages from the brain to the body’s major organs, including the heart, lungs, and intestines, and between areas of the brain that control mood, sleep, and other functions.
More recently, this therapy has been simplified by the introduction of noninvasive VNS (known as transcutaneous VNS [tVNS] ). tVNS uses a portable device to send electrical stimulation through the skin to activate the vagus nerve. Although tVNS is still experimental, the approach may offer advantages over surgical VNS, such as greater accessibility and affordability, while avoiding surgical complications.
VNS: Why it’s done
VNS was initially developed as a treatment for epilepsy. Research using brain scans showed that the procedure also can be used to improve depression symptoms.
In 2005, the FDA approved surgical VNS for depression when the following conditions are met.
- The patient is 18 years or older.
- The depression has lasted 2 or more years.
- The depression is severe or recurrent.
- The depression has not eased after trying at least four other treatments.
Despite FDA approval for depression, VNS is not intended as a first-line treatment and remains infrequently used. The results of studies examining its effectiveness for depression have been mixed. Whereas a review of clinical trials of VNS for treatment-resistant depression found a sustained reduction in depression symptoms and enhanced quality of life, other studies did not report meaningful improvements.
Although noninvasive forms of VNS have shown antidepressant effects , tVNS is not currently authorized by the FDA as a treatment for depression. However, a portable VNS device has been cleared by the FDA to treat post-traumatic stress disorder (PTSD) under a breakthrough device designation. Research is ongoing to test the efficacy and safety of tVNS for depression, PTSD, and other mental disorders.
VNS: How it works
VNS is traditionally a surgical procedure.
- A device about the size of a stopwatch called a pulse generator is implanted in the upper left side of the chest while the patient is under anesthesia.
- Connected to the pulse generator is an electrical lead wire, which goes from the generator to the left vagus nerve.
- Typically, 30-second electrical pulses are sent every 5 minutes from the generator to the vagus nerve. The duration and frequency of the pulses may vary depending on how the generator is programmed.
- The vagus nerve, in turn, delivers those electrical signals to the brain.
The pulse generator, which operates continuously, is powered by a battery that lasts around 10 years, after which it must be replaced. Patients usually do not feel pain or discomfort as the device operates.
It may be several months before noticing benefits, and not all people respond to VNS. Some patients have no improvement in symptoms, and some may get worse.
The device can be temporarily deactivated by placing a magnet over the chest where the generator is implanted. A patient may want to deactivate the device if side effects become intolerable or before engaging in strenuous activity or exercise because it can interfere with breathing. The device reactivates when the magnet is removed.
Noninvasive forms of VNS consist of a device worn around the neck or ears or a handheld device. There are many questions about the most effective stimulation sites, parameters, and protocols for tVNS, and research is ongoing to determine the optimal conditions to achieve the greatest clinical benefits.
VNS: Side effects
VNS is not without risk. There may be complications, such as infection or pain from the implant surgery, or the device may come loose, move around, or malfunction, all of which can require additional surgery to correct.
Other potential side effects include:
- Discomfort or tingling in the area where the device is implanted
- Voice changes or hoarseness
- Cough or sore throat
- Neck pain or headaches
- Breathing problems, especially during exercise
- Difficulty swallowing
- Nausea or vomiting
If cleared by the FDA, tVNS devices may help overcome some of these surgical issues. Nonetheless, mild side effects of tVNS have been reported, including:
- Tingling, pain, or itchiness around the stimulation site
- Nausea or vomiting
- Dizziness
The long-term side effects of all forms of VNS are unknown.
Experimental therapies
Other brain stimulation therapies are actively being explored for specific mental disorders. The following therapies are still considered experimental and have not yet been authorized by the FDA to treat mental disorders.
link

:max_bytes(150000):strip_icc()/GettyImages-1644727881-73ef3c9679a6447faafc74eb0ff22f1e.jpg)